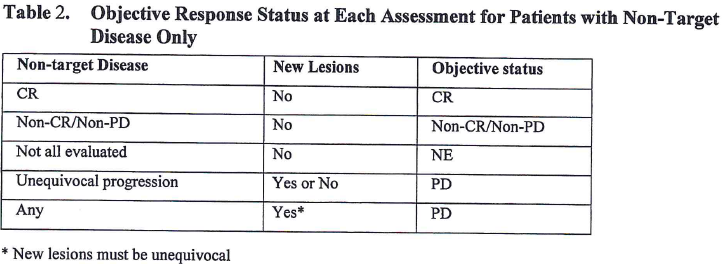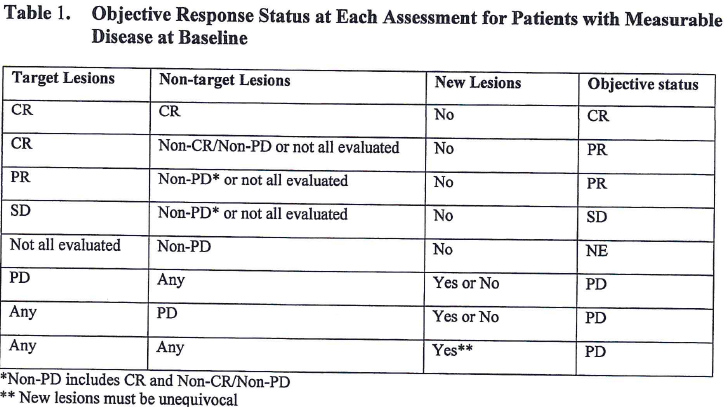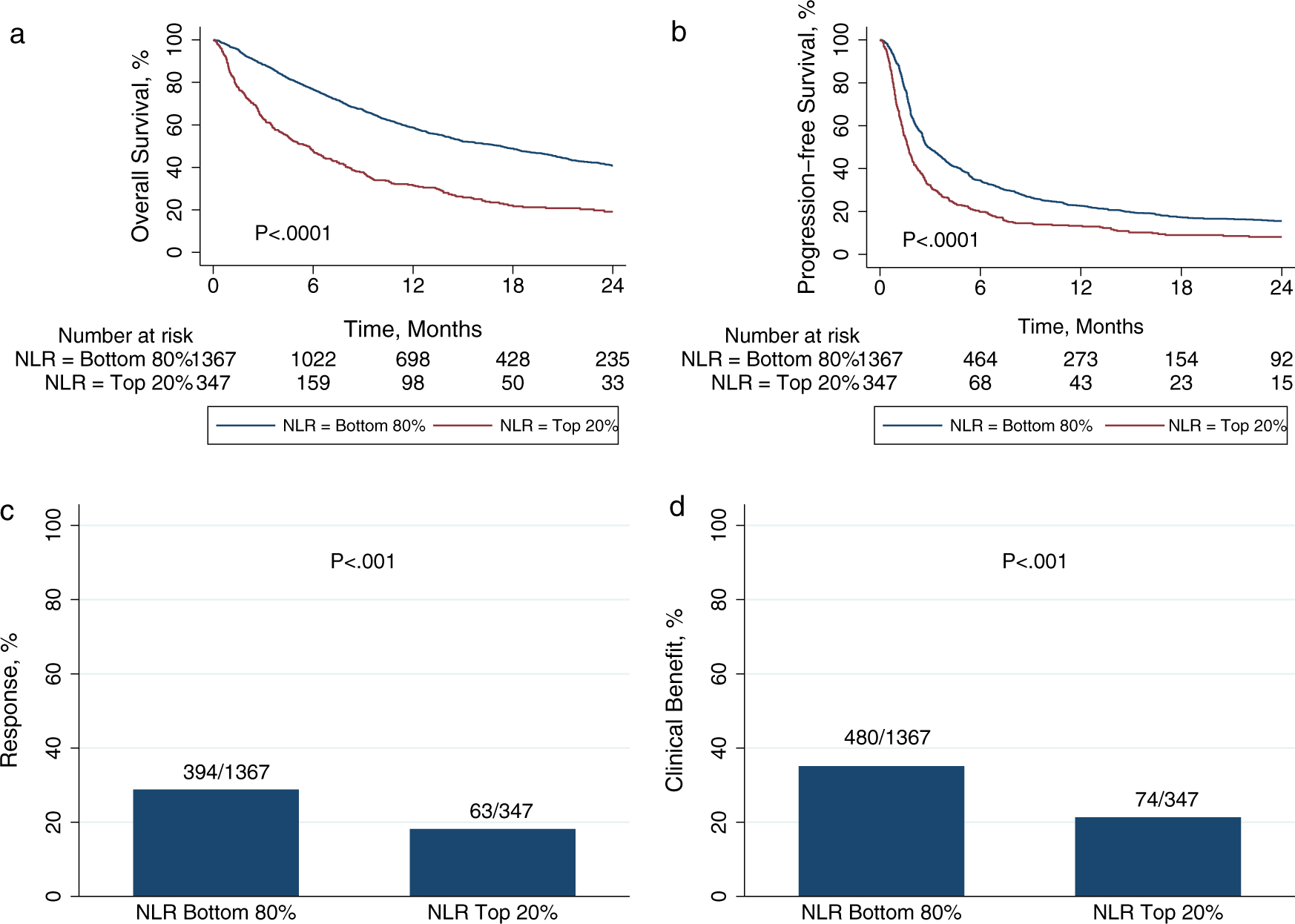
Pretreatment neutrophil-to-lymphocyte ratio and mutational burden as biomarkers of tumor response to immune checkpoint inhibitors | Nature Communications
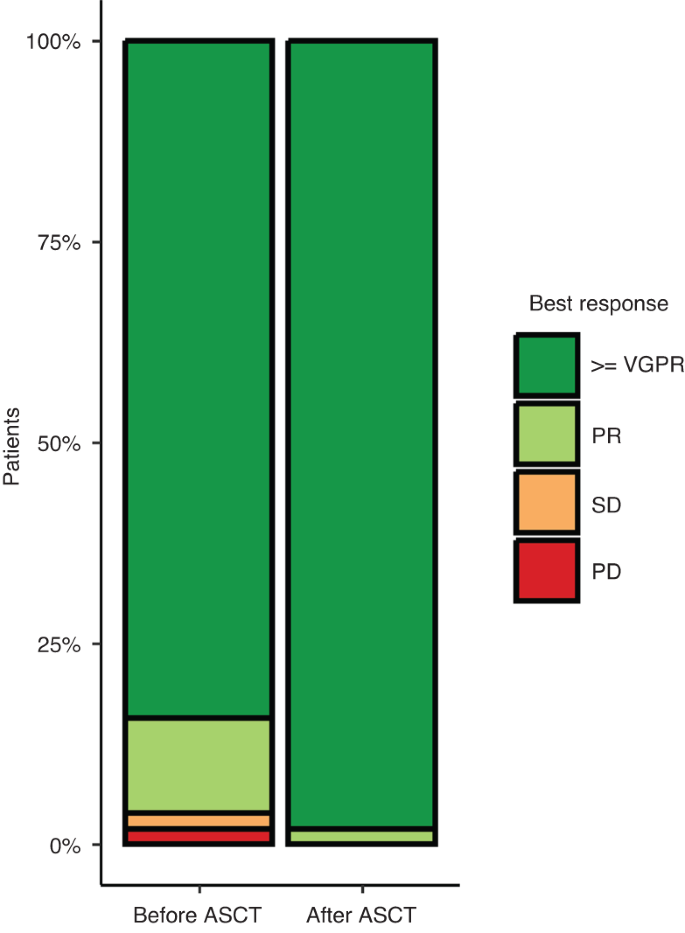
Carfilzomib, lenalidomide and dexamethasone followed by a second ASCT is an effective strategy in first relapse multiple myeloma: a study on behalf of the Chronic malignancies working party of the EBMT

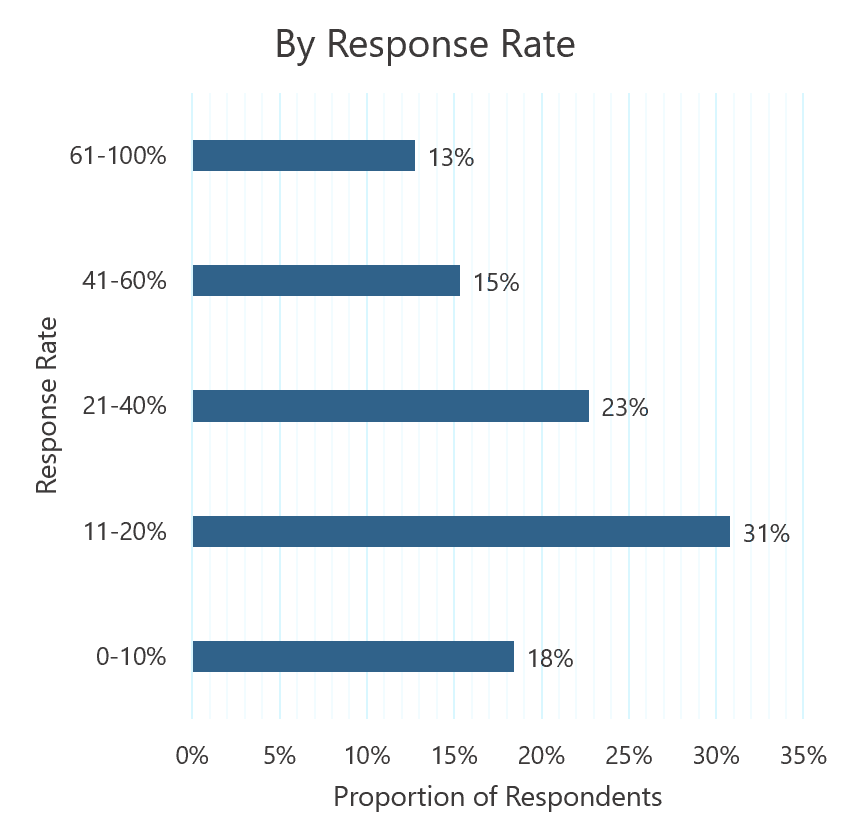
Response rates Overall response rate and depth of response according to... | Download Scientific Diagram

Objective response rate is a possible surrogate endpoint for survival in patients with advanced, recurrent ovarian cancer - ScienceDirect

Objective response rate of placebo in randomized controlled trials of anticancer medicines - eClinicalMedicine




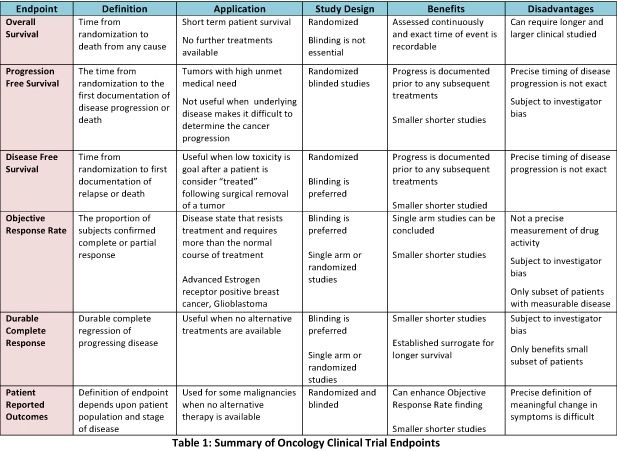
![PDF] Incorporating Clinical Trial Data Into Daily Cancer Care | Semantic Scholar PDF] Incorporating Clinical Trial Data Into Daily Cancer Care | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/30f4b9adffa198bf79a177643e74ec64b4107203/3-Table1-1.png)